Center for Nanomaterials
and Chemical Reactions
mainmenu
Group 1
Nano materials design and synthesis
- 1. Nanomaterials design & synthesis with structure-directing agents
First, a much wider landscape of structure-directing agent (SDA) molecules has to be explored in the design and synthesis of nanomaterials, as presented in Fig. 1. SDA is a key agent that can direct the structural evolution at a very early stage of the formation of the nanostructure. To date, only a limited class of SDA molecules has been exploited. For instance, most works on nanoparticle synthesis are still using commercially available surfactants as SDAs or capping agents.
In the future, the possibilities offered by exploring the vast number of organic and inorganic molecular building blocks are huge, especially considering our recent success in creating hierarchical zeolites which were achieved by altering the traditional concept of the SDA molecule. We hope to inscribe functionalities as a blueprint inside an SDA molecule so that it can generate complex levels of nano-architecture in a designed manner. We hope to build a universal design principle that can be applied to a wide range of nanomaterials. However, one of the bottlenecks in this process is the lack of knowledge about the detailed mechanisms and formation pathways of nanomaterials.
Most research efforts have thus far focused on the synthesis and basic characterization of nanostructures which appear novel rather than unveiling the detailed formation mechanisms in detail. To break through this conventional trend, we will elucidate the time-dependent reaction phenomena by employing and developing time-resolved tools such as environmental TEM and other types of modern time-resolved tools.

- 2. Chemical reaction development driven by nanocatalysts
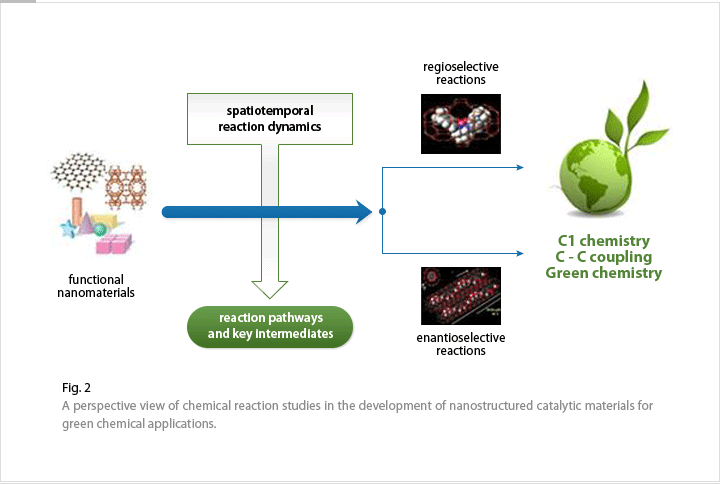
We will develop a particularly desired reaction with designed catalysis from nanostructured materials. In particular, regio- and enantio-selective chemical reactions with unprecedented reactivity and selectivity will be targeted, as pursuing this achievement is a major goal in synthetic chemistry.
Functional nanocatalysts for the desired reaction pathways can be generated by precise control of the size, shape, spatial distribution and surface morphology of the basic building blocks. We will investigate the recognition, separation, and synthesis of chemical products and thus ultimately develop a universal catalyst for chemical compound synthesis. This approach will be extended to general catalytic reactions, including C1 chemistry , C-C coupling reactions, and green chemistry (Fig.2).
- 3. Real-time atomic imaging of chemical and physical reactions
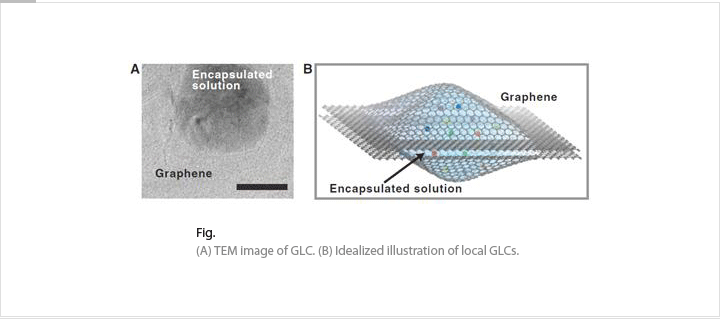
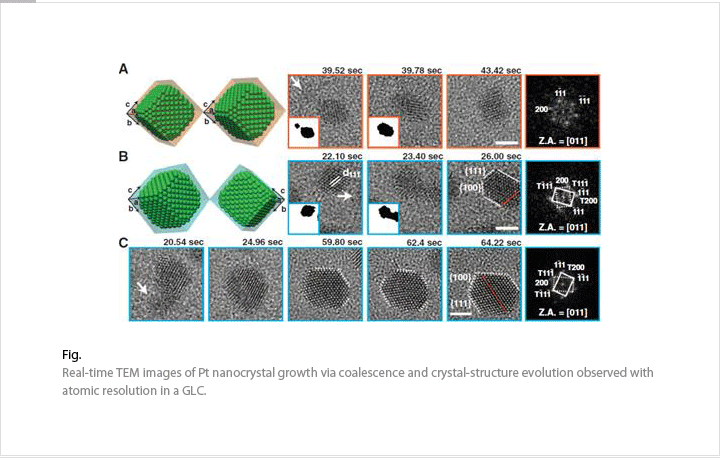
We are studying nanostructure formation mechanisms and chemical reactions in the liquid environment by combining an aberration-corrected TEM and a GLC. Atomic arrangements or compositions can be changed during chemical reactions. We will test and demonstrate nanostructured materials in real-time. These in-situ TEM experiments will help the understanding atomic movements before the industrial applications.
- 4. Atomic-scale imaging of soft nanomaterials
Transmission electron microscopy has facilitated observing morphologies, crystal structures, and compositions of various nanomaterials at an atomic level. However, soft nanomaterials have been difficult to observe using the TEM due to electron beam damage. Aberration-corrected TEM will be adopted to reduce the electron beam damage on soft nanomaterials via its operation at low voltage (< 80 keV). We will demonstrate the shape, surface morphology, and composition using the aberration-corrected TEM.