Presentation of new method for chemical reaction study
of nanomaterial in liquids
*Publication
J.M. Yuk, H.K. Seo, J.W. Choi, and J.Y. Lee, "Anisotropic Lithiation Onset in Silicon Nanoparticle Anode Revealed by In-Situ Graphene Liquid Cell Electron Microscopy", ACS Nano, 8, 7478 (2014)
Important phenomena, such as synthesis, growth, and various chemical reactions of nanomaterials occur mostly in liquid medium. In order to understand these phenomena, actual reactions of the nanomaterials in liquid at atomic level must be observed in real-time using a high spatial-and-temporal resolution transmission electron microscopy at subnanometer length and subsecond time scales. This is not easy because actual reaction conditions in liquid cannot be maintained due to evaporation inside the high-vacuum environment of the transmission electron microscope. The research team led by Jeong Yong Lee of Institute for Basic Science (IBS) Center for Nanomaterials and Chemical Reactions (Director Ryong Ryoo) developed a method for trapping liquid in the high-vacuum atmosphere with the use of graphene formed by a single layer of carbon atoms, a Graphene Liquid Cell (GLC), and also succeeded in observing the growth process of platinium(Pt) nanoparticles in liquid through a transmission electron microscope, in real-time. They further developed this method and applied it for real-time observation of lithiation reaction occurring between silicon (Si) anodeand liquid electrolyte during the charging of Li-ion battery. Moreover, they were successful in revealing the lithiation mechanism from a new perspective by overcoming the limitations that previous observationmethods have.
The research team uncovered the growth process of Pt nanoparticles in liquid by combining GLC with transmission electron microscopy (Graphene Liquid Cell–Transmission Electron Microscopy: GLC-TEM). The limitations of previous TEM studies on chemical reactions in liquid were addressed by further developing the GLC-TEM method and applying it in the observation of lithiation reaction of Li-ion batteries. They fabricated a liquid-encapsulating cell by sealing together Si nanoparticles and liquid electrolytes with graphene. Previously, complex experimental devices and methods that were used to apply the electrical bias were required for triggering the lithiation of Si nanoparticles inside TEM. The researchers eliminated that need by utilizing the electron beam from the transmission electron microscope to reduce the Li present in the electrolyte directly, which made it possible to induce and observe the reaction simultaneously.
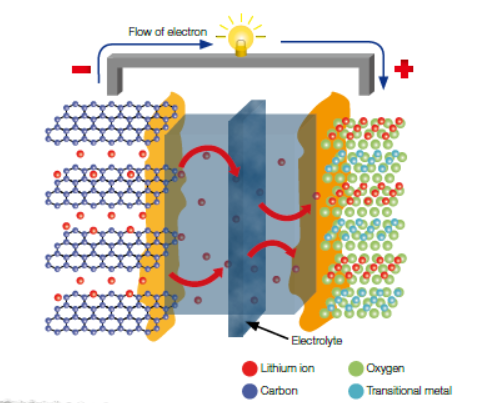
Li-ion batteries, which are widely used as power sources for small home appliances and mobile devices, are experiencing a gradual increase in demand as its usage is extending to large-scale energy storage for electric vehicles and renewable energy. Accordingly, various researchers are pushing forward to develop high-performance electrode materials, for which, an accurate understanding of the working mechanism of the corresponding material is critical. In particular, since Si has the highest theoretical energy capacity (~ 4000 mAh/g) among all materials known to date, it is receiving attention as a new-generation anode material that can replace commercially available graphite-based materials (energy capacity ~ 372 mAh/g). However, during charging and discharging, the lithiation/delithiation processes cause significant volume changes leading to severe pulverization of the electrode material, and eventually capacity degradation. The schematic diagram shows the basic structure of a Li-ion battery that is currently used ; during charging of which, the transition metals contained in the cathode are oxidized and Li+-ions are released towards and stored inside the anode material. While discharging, Li+-ions are released from the anode and they move towards and stored inside the cathode through the electrolyte.
The research team was able to track and observe a correlation between volume changes with the lithiation time of specific Si nanoparticles through the GLC-TEM method. The present study is expected to serve asan important academic foundation for designing of the next-generation Si-based anode material in Li-ion batteries. In particular, the newly pioneered real-time observation method required for battery material study has an important meaning. Furthermore, using this new GLC-TEM method, we can also expect innovative results from other interestingresearch fields concerning components in the battery , such as unveiling formation mechanism of solid-electrolyte interphase (SEI) and observing changes in quantitative distribution of lithium inside the electrode material in real-time.